
园艺学报 ›› 2023, Vol. 50 ›› Issue (5): 972-984.doi: 10.16420/j.issn.0513-353x.2022-0329
徐兰兰, 赵天孜, 张玮, 宗琛, 黄菲艺, 王建军, 侯喜林, 李英( )
)
收稿日期:2022-11-28
修回日期:2023-02-27
出版日期:2023-05-25
发布日期:2023-05-31
通讯作者:
*(E-mail:yingli@njau.edu.cn)基金资助:
XU Lanlan, ZHAO Tianzi, ZHANG Wei, ZONG Chen, HUANG Feiyi, WANG Jianjun, HOU Xilin, LI Ying( )
)
Received:2022-11-28
Revised:2023-02-27
Published:2023-05-25
Online:2023-05-31
摘要:
MAX2(MORE AXILLARY GROWTH 2)是独脚金内酯信号转导相关基因,其功能与腋芽生长有关。以白菜(Brassica campestris ssp. chinensis)分蘖菜品种‘马耳头’和普通白菜品种‘苏州青’为材料,探究MAX2对腋芽生长的影响。结果表明:从两种材料中克隆得到的BcMAX2其开放阅读框序列没有差异,‘马耳头’的BcMAX2启动子序列有2个碱基的缺失。亚细胞定位分析表明BcMAX2定位在细胞核。在拟南芥中过表达BcMAX2抑制腋芽生长,BRC1表达量升高。在‘马耳头’中沉默BcMAX2能引起BcBRC1表达量降低并促进腋芽伸长。通过对白菜cDNA文库筛选得到197个与BcMAX2互作的候选蛋白,其中多数候选互作蛋白具有结合活性和催化活性,部分候选蛋白还参与植物的信号转导和逆境响应。通过酵母双杂交和双分子荧光互补技术验证最终得到2个与BcMAX2互作的蛋白。试验表明,白菜BcMAX2可能通过促进BcBRC1的表达来负调控腋芽的生长。
中图分类号:
徐兰兰, 赵天孜, 张玮, 宗琛, 黄菲艺, 王建军, 侯喜林, 李英. 白菜(Brassica campestris ssp. chinensis)BcMAX2调控腋芽生长的功能分析[J]. 园艺学报, 2023, 50(5): 972-984.
XU Lanlan, ZHAO Tianzi, ZHANG Wei, ZONG Chen, HUANG Feiyi, WANG Jianjun, HOU Xilin, LI Ying. Function Analysis of BcMAX2 in Regulating Axillary Bud Growth in Brassica campestris ssp. chinensis[J]. Acta Horticulturae Sinica, 2023, 50(5): 972-984.
| 引物名称 | 引物序列(5′-3′) | 用途 |
|---|---|---|
| Primer name | Primer sequence | Usage |
| BcMAX2 | F:ATGGCTTCCACCACTCTCTGC | ORF扩增 |
| R:TCAGTCAATGATGATGCGGCT | Complete ORF amplification | |
| BcMAX2-P | F:TTCAACCGAATCAATCCACTATTCT | 启动子扩增 |
| R:CTCCAAGATTTGCTCTCATGGCTTCCAC | Complete promoter amplification | |
| pRI101-BcMAX2 | F:TTCTTCACTGTTGATACATATGATGGCTTCCACCACTCTCTGC | 载体构建 |
| R:TCGCCCTTGCTCACCATGGATCCTCAGTCAATGATGATGCGGCT | Construct of vector | |
| RT-BcMAX2 | F:AGGAGCTTGTGCTTGACGTT | RT-PCR |
| R:AAGACAGCGACAACAACCCT | ||
| Actin1 | F:GGAGCTGAGAGATTCCGTTG | 白菜内参基因 |
| R:GAACCACCACTGAGGACGAT | Internal reference gene in Brassica | |
| Actin2 | F:TTGACAATTGATGCAAACAATGACG | 拟南芥内参基因 |
| R:CCATTGCTTAATTCCACGGACAAAC | Internal reference genein Arabidopsis | |
| BcMAX2-BD | F:GGAATTCCATATGATGGCTTCCACCACTCTCTGC | 诱饵载体构建 |
| R:GGAATTCGTCAATGATGATGCGGCT | Construct of bait vector | |
| BcNF-YC4-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGACAACAACAACCAGCA | 载体构建 |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGAAGCAAGCTCATTACTAGT | Construct of vector | |
| BcCEBiP-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGAAACTTCCCGTTTTACCC | 载体构建 |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGGAGAAGGCATGCACAGAAC | Construct of vector | |
| BcBOLA-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGAGGAGACAGATCGTTCA GATCGTTC | 载体构建 Construct of vector |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGGATGGGAATGTCTTGATGGG | ||
| AD | F:CTATTCGATGATGAAGATACCCCACCAAACCC | cDNA插入片段扩增 |
| R:GTGAACTTGCGGGGTTTTTCAGTATCTACGATT | Amplification of cDNA fragments | |
| pCAMBIA1300-35S -NYFP-BcNF-YC4 | F:GAATTCATGGACAACAACAACCAGCA | BiFC载体构建 |
| R:GTCGACAAGCAAGCTCATTACTAGT | Construct of vector BiFC | |
| pCAMBIA1300-35S -NYFP-BcCEBiP | F:GGATCCATGGAAACTTCCCGTTTTACCC | BiFC载体构建 |
| R:ACTAGTGAGAAGGCATGCACAGAAC | Construct of vector BiFC | |
| pCAMBIA1300-35S -NYFP-BcBOLA | F:GGATCCATGGAGGAGACAGATCGTTC | BiFC载体构建 |
| R:ACTAGTGATGGGAATGTCTTGATGGG | Construct of vector BiFC | |
| pCAMBIA1300-35S -CYFP-BcMAX2 | F:GAATTCATGGCTTCCACCACTCTCTGC | BiFC载体构建 |
| R:GTCGACGGTCAATGATGATGCGGCT | Construct of vector BiFC |
表 1 PCR所用引物序列
Table 1 Primer sequences of PCR amplification
| 引物名称 | 引物序列(5′-3′) | 用途 |
|---|---|---|
| Primer name | Primer sequence | Usage |
| BcMAX2 | F:ATGGCTTCCACCACTCTCTGC | ORF扩增 |
| R:TCAGTCAATGATGATGCGGCT | Complete ORF amplification | |
| BcMAX2-P | F:TTCAACCGAATCAATCCACTATTCT | 启动子扩增 |
| R:CTCCAAGATTTGCTCTCATGGCTTCCAC | Complete promoter amplification | |
| pRI101-BcMAX2 | F:TTCTTCACTGTTGATACATATGATGGCTTCCACCACTCTCTGC | 载体构建 |
| R:TCGCCCTTGCTCACCATGGATCCTCAGTCAATGATGATGCGGCT | Construct of vector | |
| RT-BcMAX2 | F:AGGAGCTTGTGCTTGACGTT | RT-PCR |
| R:AAGACAGCGACAACAACCCT | ||
| Actin1 | F:GGAGCTGAGAGATTCCGTTG | 白菜内参基因 |
| R:GAACCACCACTGAGGACGAT | Internal reference gene in Brassica | |
| Actin2 | F:TTGACAATTGATGCAAACAATGACG | 拟南芥内参基因 |
| R:CCATTGCTTAATTCCACGGACAAAC | Internal reference genein Arabidopsis | |
| BcMAX2-BD | F:GGAATTCCATATGATGGCTTCCACCACTCTCTGC | 诱饵载体构建 |
| R:GGAATTCGTCAATGATGATGCGGCT | Construct of bait vector | |
| BcNF-YC4-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGACAACAACAACCAGCA | 载体构建 |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGAAGCAAGCTCATTACTAGT | Construct of vector | |
| BcCEBiP-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGAAACTTCCCGTTTTACCC | 载体构建 |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGGAGAAGGCATGCACAGAAC | Construct of vector | |
| BcBOLA-AD | F:CATATGGCCATGGAGGCCAGTGAATTCATGGAGGAGACAGATCGTTCA GATCGTTC | 载体构建 Construct of vector |
| R:ATCTGCAGCTCGAGCTCGATGGATCCGGGATGGGAATGTCTTGATGGG | ||
| AD | F:CTATTCGATGATGAAGATACCCCACCAAACCC | cDNA插入片段扩增 |
| R:GTGAACTTGCGGGGTTTTTCAGTATCTACGATT | Amplification of cDNA fragments | |
| pCAMBIA1300-35S -NYFP-BcNF-YC4 | F:GAATTCATGGACAACAACAACCAGCA | BiFC载体构建 |
| R:GTCGACAAGCAAGCTCATTACTAGT | Construct of vector BiFC | |
| pCAMBIA1300-35S -NYFP-BcCEBiP | F:GGATCCATGGAAACTTCCCGTTTTACCC | BiFC载体构建 |
| R:ACTAGTGAGAAGGCATGCACAGAAC | Construct of vector BiFC | |
| pCAMBIA1300-35S -NYFP-BcBOLA | F:GGATCCATGGAGGAGACAGATCGTTC | BiFC载体构建 |
| R:ACTAGTGATGGGAATGTCTTGATGGG | Construct of vector BiFC | |
| pCAMBIA1300-35S -CYFP-BcMAX2 | F:GAATTCATGGCTTCCACCACTCTCTGC | BiFC载体构建 |
| R:GTCGACGGTCAATGATGATGCGGCT | Construct of vector BiFC |

图2 ‘苏州青’和‘马耳头’中BcMAX2启动子序列的差异 ATG为BcMAX2起始密码子。
Fig. 2 Difference in promoter of BcMAX2 sequences between‘Maertou’and‘Suzhouqing’ The ATG is the BcMAX2 start codon.
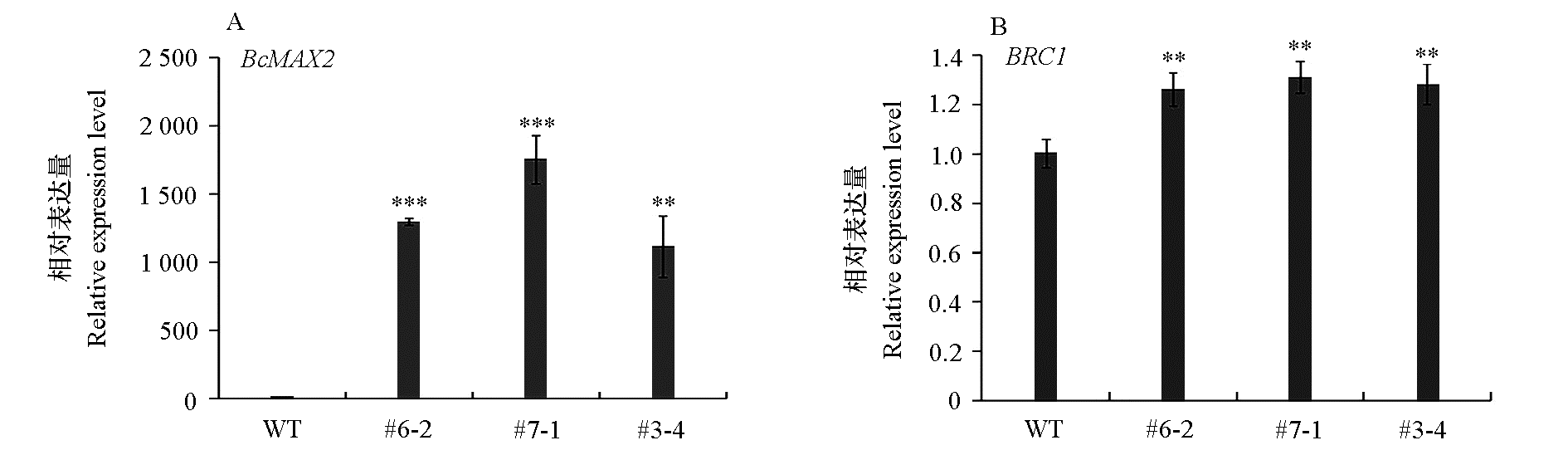
图6 拟南芥野生型(WT)和过表达植株(#6-2、#7-1和 #3-4)BcMAX2以及BRC1的表达量
Fig. 6 Expression level of BcMAX2 and BRC1 in wild-type(WT)and overexpressed(#6-2,#7-1 and #3-4)plants ** P < 0.01,*** P < 0.001.
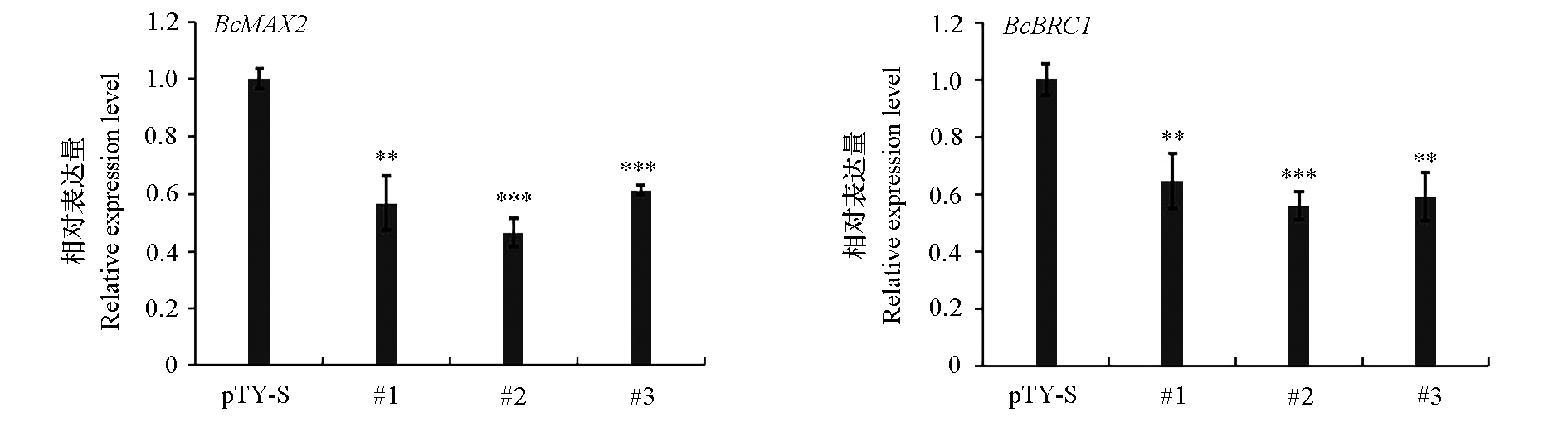
图8 白菜侵染pTY-S和pTY-BcMAX2(#1、#2和#3)植株BcMAX2和BcBRC1的表达量
Fig. 8 Expression of BcMAX2 and BcBRC1 after being infected with pTY-S and pTY-BcMAX2(#1,#2 and #3) ** P < 0.01,*** P < 0.001.
| [1] |
Aguilar-Martínez J A, Poza-Carrión C, Cubas P. 2007. Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds. The Plant Cell, 19 (2):458-472.
doi: 10.1105/tpc.106.048934 URL |
| [2] |
Arite T, Umehara M, Ishikawa S, Hanada A, Maekawa M, Yamaguchi S, Kyozuka J. 2009. d14,a strigolactone-insensitive mutant of rice,shows an accelerated outgrowth of tillers. Plant and Cell Physiology, 50 (8):1416-1424.
doi: 10.1093/pcp/pcp091 URL |
| [3] |
Bennett T, Sieberer T, Willett B, Booker J, Luschnig C, Leyser O. 2006. The Arabidopsis MAX pathway controls shoot branching by regulating auxin transport. Current Biology, 16 (6):553-563.
doi: 10.1016/j.cub.2006.01.058 pmid: 16546078 |
| [4] | Bian Hai-yan, Zhong Qi-wen, Huang Si-jie, Wang Li-hui, Yang Shi-peng, Tian Jie. 2018. Analysis of gene expressions of fructan metabolism key enzymes in garlic under drought stress. Molecular Plant Breeding, 16 (20):6770-6776. (in Chinese) |
| 边海燕, 钟启文, 黄思杰, 王丽慧, 杨世鹏, 田洁. 2018. 干旱胁迫下大蒜果聚糖代谢关键酶基因的表达分析. 分子植物育种, 16 (20):6770-6776. | |
| [5] |
Booker J, Sieberer T, Wright W, Williamson L, Willett B, Stirnberg P, Turnbull C, Srinivasan M, Goddard P, Leyser O. 2005. MAX1 encodes a cytochrome P 450 family member that acts downstream of MAX3/4 to produce a carotenoid-derived branch-inhibiting hormone. Developmental Cell, 8 (3):443-449.
doi: 10.1016/j.devcel.2005.01.009 URL |
| [6] |
Braun N, Germain A D S, Pillot J P, Stephanie B M, Dalmais M, Antoniadi I, Li X, Alessandra M G, Signor C L, Bouteiller N, Luo D, Bendahmane A, Turnbull C, Rameau C. 2012. The pea TCP transcription factor PsBRC 1 acts downstream of strigolactones to control shoot branching. Plant Physiology, 158 (1):225-238.
doi: 10.1104/pp.111.182725 pmid: 22045922 |
| [7] |
Carolien R S, Salim A B, Krol S D V, Bouwmeester H. 2013. The biology of strigolactones. Trends in Plant Science, 18 (2):72-83.
doi: 10.1016/j.tplants.2012.10.003 URL |
| [8] | Cao Xue-wei. 2016. The formation mechanism and candidate gene identification of tillering in non-heading Chinese cabbage[Ph. D. Dissertation]. Nanjing: Nanjing Agricultural University. (in Chinese) |
| 曹学伟. 2016. 不结球白菜分枝性状的发生机理及其候选基因挖掘[博士论文]. 南京: 南京农业大学. | |
| [9] |
Chen X M, Shi X Y, Ai Q, Han J Y, Wang H S, Fu Q S. 2022. Transcriptomic and metabolomic analyses reveal that exogenous strigolactones alleviate the response of melon root to cadmium stress. Horticultural Plant Journal, 8 (5):637-649.
doi: 10.1016/j.hpj.2022.07.001 URL |
| [10] |
Cheng X, Ruyter-Spira C, Bouwmeester H. 2013. The interaction between strigolactones and other plant hormones in the regulation of plant development. Frontiers in Plant Science, 4:199.
doi: 10.3389/fpls.2013.00199 pmid: 23785379 |
| [11] | Cui Hong-mi. 2016. Study on the main influence factors of non-heading Chinese cabbage tillering[M. D. Dissertation]. Nanjing: Nanjing Agricultural University. (in Chinese) |
| 崔红米. 2016. 影响不结球白菜分枝性状的主要因素探讨[硕士论文]. 南京: 南京农业大学. | |
| [12] | Domagalska M A, Leyser O. 2011. Signal integration in the control of shoot branching. Nature Reviews. Molecular Cell Biology, 12 (4):211-221. |
| [13] |
Ferrero M, Pagliarani C, Novák O, Ferrandino A, Cardinale F, Visentin I. 2018. Exogenous strigolactone interacts with abscisic acid-mediated accumulation of anthocyanins in grapevine berries. J Exp Bot, 69:2391-2401.
doi: 10.1093/jxb/ery033 pmid: 29401281 |
| [14] |
Gomez-Roldan V, Fermas S, Brewer P B, Puech-Pagès V, Dun E A, Pillot J P, Letisse F, Matusova R, Danoun S, Portais J C. 2008. Strigolactone inhibition of shoot branching. Nature, 455 (7210):189-194.
doi: 10.1038/nature07271 |
| [15] |
Hamiaux C, Drummond R S M, Janssen B J, Ledger S E, Cooney J M, Newcomb R D, Snowden K C. 2012. DAD2 is an α/β hydrolase likely to be involved in the perception of the plant branching hormone,strigolactone. Current Biology, 22 (21):2032-2036.
doi: 10.1016/j.cub.2012.08.007 pmid: 22959345 |
| [16] | Hou Xilin, Li Ying, Huang Feiyi. 2020. Advances in molecular biology of main characters and breeding technology in non-heading Chinese cabbage (Brassica campestris ssp. chinensis). Acta Horticulturae Sinica, 47 (9):1663-1677. (in Chinese) |
| 侯喜林, 李英, 黄菲艺. 2020. 不结球白菜(Brassica campestris ssp. chinensis)主要性状及育种技术的分子生物学研究新进展. 园艺学报, 47 (9):1663-1677. | |
| [17] | Hu J, Ji Y Y, Hu X T, Sun S Y, Wang X L. 2020. BES1 functions as the co-regulator of D53-like SMXLs to inhibit BRC1 expression in strigolactone-regulated shoot branching in Arabidopsis. Plant Communications, 1 (3):30-41. |
| [18] |
Jiang L, Liu X, Xiong G S, Liu H H, Chen F L, Wang L, Meng X B, Liu G F, Hong Y, Yuan Y D, Yi W, Zhao L H, Ma H L, He Y Z, Wu H S, Melcher K, Qian Q, Xu H E, Wang Y H, Li J Y. 2013. DWARF 53 acts as a repressor of strigolactone signalling in rice. Nature, 504 (7480):401-405.
doi: 10.1038/nature12870 |
| [19] |
Johnson X, Brcich T, Dun E A, Goussot M, Haurogné K, Beveridge C A, Rameau C. 2006. Branching genes are conserved across species. Genes controlling a novel signal in pea are coregulated by other long-distance signals. Plant Physiology, 142 (3):1014-1026.
doi: 10.1104/pp.106.087676 pmid: 16980559 |
| [20] | Li Ju. 2015. Expression analysis of TCP family transcription factor genes BcBRC1 and tillering traits genetic analysis in non-heading Chinese cabbage[M. D. Dissertation]. Nanjing: Nanjing Agricultural University. (in Chinese) |
| 李菊. 2015. 不结球白菜TCP家族转录因子基因BcBRC1的表达分析及分枝性状遗传分析[硕士论文]. 南京: 南京农业大学. | |
| [21] | Li Z D, Li Y, Liu T K, Zhang C W, Xiao D, Hou X L. 2022. Non-heading Chinese cabbage database:an open-access platform for the genomics of Brassica campestris(syn. Brassica rapa)ssp. chinensis. Plants, 11 (8):101596181. |
| [22] | Seale M, Bennett T, Leyser O. 2017. BRC1 expression regulates bud activation potential but is not necessary or sufficient for bud growth inhibition in Arabidopsis. Development, 144 (9):1661-1673. |
| [23] |
Shen R X, Ma X L, Wang H Y. 2020. SMXL6/7/8 :Dual-function transcriptional repressors of strigolactone signaling. Molecular Plant, 13 (9):1244-1246.
doi: 10.1016/j.molp.2020.08.002 URL |
| [24] |
Snowden K C, Simkin A J, Janssen B J, Templeton K R, Loucas H M, Simons J L, Karunairetnam S, Gleave A P, Klee C H J. 2005. The DECREASED APICAL DOMINANCE1/Petunia hybrida CAROTENOID CLEAVAGE DIOXYGENASE8 gene affects branch production and plays a role in leaf senescence,root growth,and flower development. The Plant cell, 17 (3):746-759.
doi: 10.1105/tpc.104.027714 URL |
| [25] |
Stirnberg P, Furner I J, Leyseret H M O. 2007. MAX2 participates in an SCF complex which acts locally at the node to suppress shoot branching. The Plant Journal, 50 (1):80-94.
doi: 10.1111/j.1365-313X.2007.03032.x URL |
| [26] |
Stirnberg P, van de Sande K, Leyser H M. 2002. MAX1 and MAX2 control shoot lateral branching in Arabidopsis. Development, 129 (5):1131-1141.
doi: 10.1242/dev.129.5.1131 pmid: 11874909 |
| [27] |
Soundappan I, Bennett T, Morffy N, Liang Y, Nelson D C. 2015. SMAX1-LIKE/D 53 family members enable distinct MAX2-dependent responses to strigolactones and karrikins in Arabidopsis. The Plant Cell, 27 (11):3143-3159.
doi: 10.1105/tpc.15.00562 pmid: 26546447 |
| [28] |
Sun W J, Ji X L, Song L Q, Wang X F, You C X, Hao Y J. 2021. Functional identification of MdSMXL8.2,the homologous gene of strigolactones pathway repressor protein gene in Malus × domestica. Horticultural Plant Journal, 7 (4):275-285.
doi: 10.1016/j.hpj.2021.01.001 URL |
| [29] |
Umehara M, Hanada A, Yoshida S, Akiyama K, Arite T, Noriko T K, Magome H, Kamiya Y, Shirasu K, Yoneyama K, Kyozuka J, Yamaguchi S. 2008. Inhibition of shoot branching by new terpenoid plant hormones. Nature, 455 (7210):195-200.
doi: 10.1038/nature07272 |
| [30] |
Wang L, Wang B, Jiang L, Liu X, Li J Y. 2015. Strigolactone signaling in Arabidopsis regulates shoot development by targeting D53-Like SMXL repressor proteins for ubiquitination and degradation. The Plant cell, 27 (11):3128-3142.
doi: 10.1105/tpc.15.00605 pmid: 26546446 |
| [31] |
Wang L, Wang B, Yu H, Guo H Y, Lin T, Kou L Q, Wang A Q, Shao N, Ma H Y, Xiong G S, Li X Q, Yang J, Chu J F, Li J Y. 2020. Transcriptional regulation of strigolactone signalling in Arabidopsis. Nature, 583 (7815):277-281.
doi: 10.1038/s41586-020-2382-x |
| [32] |
Wang H W, Chen W X, Eggert K, Charnikhova T, Bouwmeester H, Schweizer P, Hajirezaei M R, Seiler C, Sreenivasulu N, Wirén N V, Kuhlmann M. 2018. Abscisic acid influences tillering by modulation of strigolactones in barley. Journal of Experimental Botany, 69 (16):3883-3898.
doi: 10.1093/jxb/ery200 pmid: 29982677 |
| [33] |
Waters M T, Brewer P B, Bussel J D, Smith S M, Beveridge C A. 2012. The Arabidopsis ortholog of rice DWARF27 acts upstream of MAX1 in the control of plant development by strigolactones. Plant Physiology, 159 (3):1073-1085.
doi: 10.1104/pp.112.196253 pmid: 22623516 |
| [34] | Yang Xue-dong, Dai Wei, Zhang Chang-wei, Wang Jin-yan, Kong Min, Hou Xi-lin. 2012. The technology system establishment of VIGS in non-heading Chinese cabbage. Acta Horticulturae Sinica, 39 (11):2168-2174. (in Chinese) |
| 杨学东, 戴薇, 张昌伟, 王金彦, 孔敏, 侯喜林. 2012. 白菜病毒诱导基因沉默技术体系的建立. 园艺学报, 39 (11):2168-2174. | |
| [35] | Zhang Wei, Guo Ming-liang, Huang Fei-yi, Long Yan, Hou Xi-lin, Li Ying. 2021. Homologous cloning and functional analysis of the tillering regulation gene BcMAX1 in non-heading Chinese cabbage. Journal of Nanjing Agricultural University, 44 (2):241-248. (in Chinese) |
| 张玮, 郭明亮, 黄菲艺, 龙言, 侯喜林, 李英. 2021. 不结球白菜分枝调控基因BcMAX1的同源克隆及功能分析. 南京农业大学学报, 44 (2):241-248. | |
| [36] |
Zou J H, Zhang S Y, Zhang W P, Li G, Chen Z X, Zhai W X, Zhao X F, Pan X B, Xie Q, Zhu L H. 2006. The rice HIGH‐TILLERING DWARF 1 encoding an ortholog of Arabidopsis MAX3 is required for negative regulation of the outgrowth of axillary buds. The Plant Journal, 48 (5):687-698.
doi: 10.1111/tpj.2006.48.issue-5 URL |
| [1] | 李仁杰 , 汪承刚 , 张胜男 , 单国雷 , 陈国户 , 侯金锋 , 黄兴学 , 袁凌云 , . 耐热不结球白菜品种‘夏抗 718’ [J]. 园艺学报, 2023, 50(S1): 43-44. |
| [2] | 韩书辉, 韩彩锋, 韩书荣, 韩彩梅, 韩 旭. 秋大白菜新品种‘胶研秋宝’[J]. 园艺学报, 2022, 49(S2): 83-84. |
| [3] | 汪维红, 张凤兰, 余阳俊, 张德双, 赵岫云, 于拴仓, 苏同兵, 李佩荣, 辛晓云. 秋大白菜新品种‘京秋1518’[J]. 园艺学报, 2022, 49(S2): 85-86. |
| [4] | 余阳俊, 汪维红, 苏同兵, 张凤兰, 张德双, 赵岫云, 于拴仓, 李佩荣, 辛晓云, 王 姣. 抗根肿病耐抽薹大白菜新品种‘京春CR3’[J]. 园艺学报, 2022, 49(S2): 87-88. |
| [5] | 王丽丽, 王 鑫, 吴海东, 温 蔷, 杨晓飞. 抗根肿病大白菜新品种‘辽白28’[J]. 园艺学报, 2022, 49(S2): 89-90. |
| [6] | 余阳俊, 苏同兵, 张凤兰, 张德双, 赵岫云, 于拴仓, 汪维红, 李佩荣, 辛晓云, 王 姣, 武长见. 紫色苗用型大白菜新品种‘京研紫快菜’[J]. 园艺学报, 2022, 49(S2): 91-92. |
| [7] | 黄 鹂, 陈财志, 余小林, 姚祥坦, 曹家树, . 早中熟普通白菜新品种‘浙大青’[J]. 园艺学报, 2022, 49(S2): 93-94. |
| [8] | 徐立功, 韩太利, 孙继峰, 杨晓东, 谭金霞. 苗用大白菜新品种‘锦绿2号’[J]. 园艺学报, 2022, 49(S1): 67-68. |
| [9] | 邵贵荣, 朱 彬, 林 晓, 曹 萍, 方 勇, 崔 田, 蒋 鹏, 林咏铭, 林 魁, 林志滔. 白菜新品种‘金品008’[J]. 园艺学报, 2022, 49(S1): 69-70. |
| [10] | 姜悦悦, 王田田, 赵 阳, 汪承刚, 侯金锋, 袁凌云. 白菜新品种‘皖绿2号’[J]. 园艺学报, 2022, 49(S1): 71-72. |
| [11] | 王钰, 张雪, 张学颖, 张思雨, 闻婷婷, 王迎君, 甘彩霞, 庞文星. 抗毒素Camalexin对大白菜抗根肿病的作用研究[J]. 园艺学报, 2022, 49(8): 1689-1698. |
| [12] | 张鲁刚, 卢倩倩, 何琼, 薛一花, 马晓敏, 马帅, 聂姗姗, 杨文静. 紫橙色大白菜新种质的创制[J]. 园艺学报, 2022, 49(7): 1582-1588. |
| [13] | 陈道宗, 刘镒, 沈文杰, 朱博, 谭晨. 白菜、甘蓝和甘蓝型油菜PAP1/2同源基因的鉴定及分析[J]. 园艺学报, 2022, 49(6): 1301-1312. |
| [14] | 王光鹏, 刘同坤, 徐新凤, 李竹帛, 高瞻远, 侯喜林. 大白菜LEA家族基因的鉴定及其部分成员在低温胁迫下的表达分析[J]. 园艺学报, 2022, 49(2): 304-318. |
| [15] | 王荣花, 王树彬, 刘栓桃, 李巧云, 张志刚, 王立华, 赵智中. 大白菜花茎蜡粉近等基因系转录组分析[J]. 园艺学报, 2022, 49(1): 62-72. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
版权所有 © 2012 《园艺学报》编辑部 京ICP备10030308号-2 国际联网备案号 11010802023439
编辑部地址: 北京市海淀区中关村南大街12号中国农业科学院蔬菜花卉研究所 邮编: 100081
电话: 010-82109523 E-Mail: yuanyixuebao@126.com
技术支持:北京玛格泰克科技发展有限公司